Overview
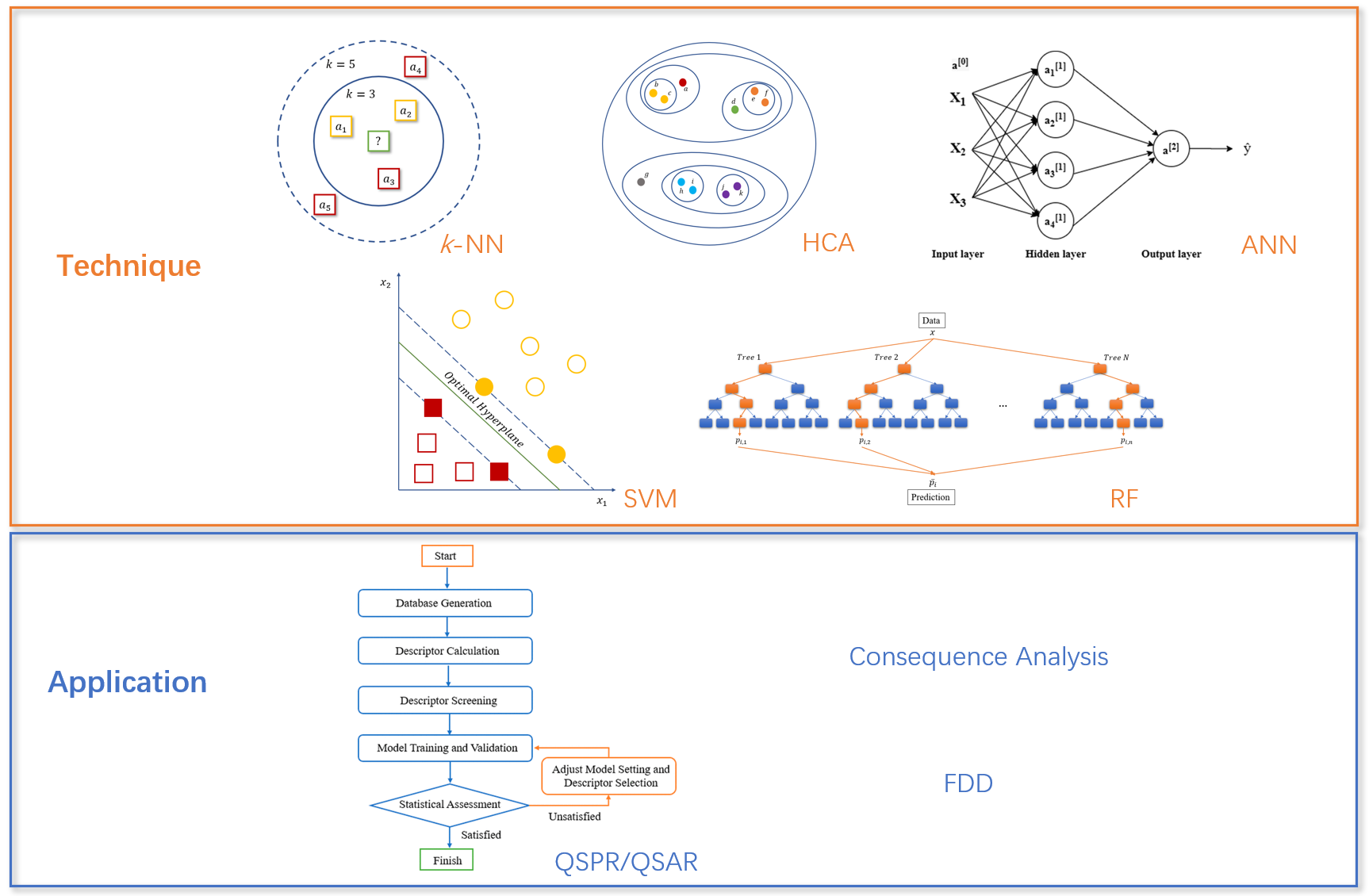
Machine learning (ML) and deep learning (DL) are a subset of artificial intelligence (AI) that can automatically learn from data and can perform tasks such as predictions for decision-making. Interdisciplinary studies combining ML/DL with chemical health and safety have demonstrated their unparalleled advantages in identifying trend and prediction assistance, which can greatly save manpower, material resources, and financial resources.
We have published over 80 peer-reviewed journal papers and 3 invited reviews in this emerging field. A new book titled “Machine Learning in Chemical Safety and Health: Fundamentals with Applications” has been published by the Wiley.
Research Objective
To apply AI in process safety, including both machine learning and deep learning techniques to:
- Predict material/molecular properties for
- material design
- chemical discovery
- chemical safety
- Predict process conditions in process engineering for
- fault detection
- process safety management (PSM)
- process incident prevention
Research Method
- Integrate Computational Chemistry with Machine Learning
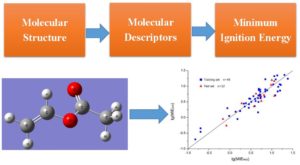
Molecular modeling can be combined with quantitative structure–property relationship (QSPR) models to predict flammability of hydrocarbons. Minimum ignition energy (MIE) is one of the most important parameters when characterizing probability of ignition. For the first time, my research group has proposed two QSPR models based on existing experimental data. Both models are validated to have excellent performances and hence are qualified to predict MIE values for chemicals with no experimental data available. These two validated models can also help gain a better understanding of effects of molecular structures on ignition properties of hydrocarbon fuels. This research provides general guideline and methodology of establishing QSPR models to predict hazardous properties of chemicals. The result was published in Industrial & Engineering Chemistry Research and was featured by the magazine Advances in Engineering.
- Integrate Computational Fluid Dynamics (CFD) with Deep Learning
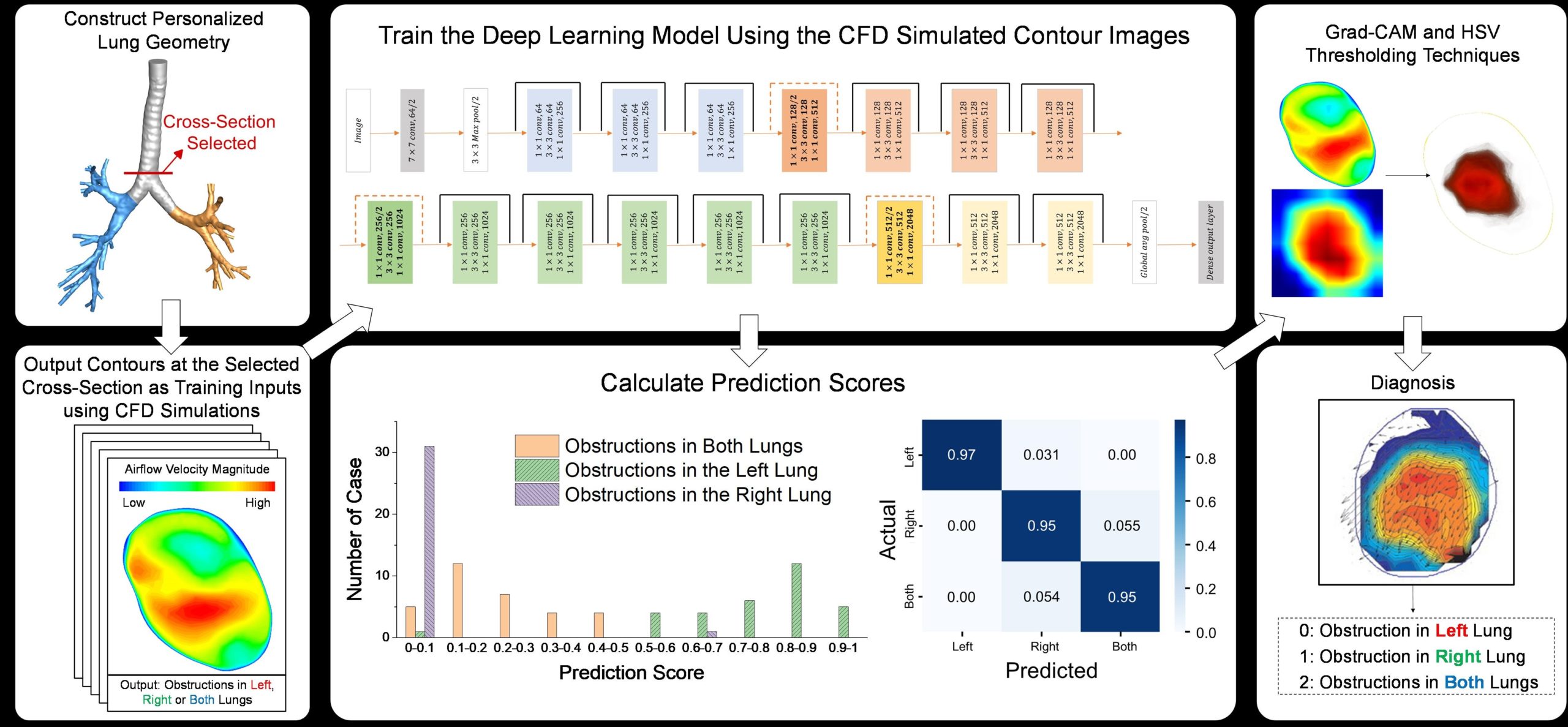
Expiratory airflow velocity contours were obtained from CFD simulations in a subject-specific 3D tracheobronchial tree. CNN-based model results indicate that strong relevance exists between the locations of the obstruction and the expiratory airflow velocity distributions at selected cross-sections in the trachea. This deep learning study provides a feasible way to identify early-stage distal airway obstruction, provide a noninvasive and effective diagnostic tool with a classification model to effectively diagnose COPDs at their early stage, and provide detailed information for precise treatment with better therapeutic outcomes. The result was published in Journal of Fluids Engineering.
Selected Publications
(1) Thermal Stability of Metal–Organic Frameworks (MOFs): Concept, Determination, and Model Prediction Using Computational Chemistry and Machine Learning, Industrial & Engineering Chemistry Research 2022, https://doi.org/10.1021/acs.iecr.2c00561.
(2) Development of Solubility Prediction Models with Ensemble Learning, Industrial & Engineering Chemistry Research 2021, https://doi.org/10.1021/acs.iecr.1c02142.
(3) Machine Learning and Deep Learning in Chemical Health and Safety: A Systematic Review of Techniques and Applications, ACS Chemical Health & Safety, 2020, 27 (6), 316–334. 10.1021/acs.chas.0c00075
(4) Review of Recent Developments of Quantitative Structure-Property Relationship Models on Fire and Explosion Related Properties, Process Safety and Environmental Protection, 2019, 129, 280-290.
(5) Prediction of Minimum Ignition Energy from Molecular Structure Using Quantitative Structure-Property Relationship (QSPR) Models, Industrial & Engineering Chemistry Research 2017, 56 (1), 47–51.
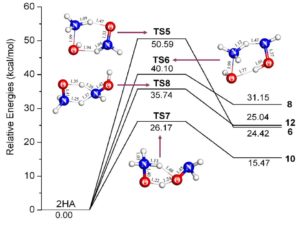
(6) Thermal decomposition pathways of hydroxylamine: theoretical investigation on the initial steps, Journal of Physical Chemistry A 2010, 114 (34), 9262-9269.